“It works!” trumpeted the normally stoic Nature journal about Pfizer’s early release results in a Phase III trial of its vaccine for COVID-19. Pfizer stated the vaccine was 90% effective when trial participants were exposed to SARS CoV-2, the virus said to cause COVID-19 symptoms.
Not so fast. Pfizer’s study protocol states cases count even if a trial participant has a positive test and only one symptom — like a cough, chills or diarrhea — that could easily be caused by one of 1,400 human pathogens, including 200 viruses known to infect humans. Except for Hepatitis A, B, C and HIV, the study protocol is silent on testing for other infectious causes of the participant’s symptoms.
In other words, the study suffers from confirmation bias. A cough and a positive test equals COVID, even when an “alternative possible diagnosis” could be the real cause of the symptoms. Absence of evidence is not evidence of absence.
Pfizer, partnering with BioNTech, uses new messenger RNA (mRNA) technology in their vaccine, and will apply for a coveted Emergency Use Authorization from the U.S. Food and Drug Administration (FDA), likely before the end of November. Pfizer’s announcement comes after 94 trial participants — of more than 43,000 — tested positive for SARS CoV-2. Pfizer has not released data on whether the cases were mild, moderate or severe.
Early results may sound encouraging to some, but experts like Eric Topol, director of the Scripps Research Translational Institute, indicated “the vaccine may not turn out to be as effective once the trial is complete and all the data has been analysed [sic],” however “its effectiveness is likely to stay well above 50%.” Pfizer’s study protocol (p. 103) states “success at the final analysis” will achieve a minimum 30% efficacy, with greater than 98% certainty. We hope the vaccine is not 30 to 90% effective against mild illness.
Will the Pfizer vaccine work, and is it safe? Questions remain. Any vaccine, including all COVID-19 vaccines, must be proven both safe and effective before being administered to high-risk groups or to the global population.
A successful vaccine must prevent severe illness, hospitalizations or death, without serious adverse events that outweigh these benefits. It cannot just claim to prevent mild cases that would resolve on their own. It must also prevent person-to-person transmission. The trial data must be sufficiently powered to answer these questions, not just for the healthy, but for high-risk groups such as the elderly and those with underlying conditions.
Any initial protection from COVID-19 symptoms should also endure, and not wane after a few weeks or a few months. Questions remain about these valid criteria as applied to COVID-19 vaccine candidates. Recent publications in leading medical journals indicate that the answers to safety and efficacy questions may be less than assuring.
As most of the world still suffers under lockdowns, many wonder if these magic keys dangled to unlock us — the vaccines being developed for COVID-19 — will cause more injuries and deaths than those caused by COVID-19 illness.
Illegal to mandate vaccines under Emergency Use Authorization
Any COVID vaccine(s) approved for emergency use should be voluntary, since the vaccine(s) are considered investigational and are held to a much lower standard for both efficacy and safety. For example, compared to the non-emergency approval process to get full licensure, an emergency approval allows for a vaccine that “may” be effective, compared to the non-emergency approval process where a vaccine must demonstrate “substantial” effectiveness.
Emergency Use Authorization (EUA) law is clear: States are barred from mandating a vaccine approved for emergency usage. (See Section VI. Preemption.) It also should be illegal for private businesses, airlines or your employer to mandate a vaccination while it is approved under an EUA.
The New York Bar Association somehow missed this materially important barrier to mandates. Their attorneys published a position statement urging states to make COVID vaccination mandatory, allowing only a medical exemption. It appears that these lawyers either have supernatural foresight that COVID vaccines will be granted full licensure sometime in the future, or they have great hubris thinking they can override EUA law.
Only if the FDA were to grant full licensure, which normally takes years, would the states or businesses be allowed to consider vaccine mandates. The PREP Act exempts COVID vaccine manufacturers from liability, even if the vaccine(s) harm recipients, so the idea of mandates is particularly frightening.
Settling for a ‘new normal’
Public health authorities tantalize us with the idea of a “new normal” after a vaccine for COVID-19 is widely available. The term implies, and has been widely interpreted to mean, that for society to return to normal, the vaccine would prevent person-to-person transmission so everything could “open up.”
As appealing as this may sound to those harmed by lockdowns, only 42% of Americans now say they would get a COVID vaccine, according to the latest YouGov poll. Even among this sanguine minority, two-thirds harbor concerns about COVID vaccine safety. If public health authorities want high uptake of the vaccine, they need to push manufacturers to provide transparent trial information to address concerns, as the vaccine will have to be voluntary if any EUAs are granted.
In June, vaccine industry spokesperson Dr. Peter Hotez said, “Ideally, you want an antiviral vaccine to do two things. … First, reduce the likelihood you will get severely ill and go to the hospital, and two, prevent infection and therefore interrupt disease transmission.”
However, last week, Dr. Anthony Fauci, director of the National Institute of Allergy and Infectious Diseases (NIAID) and White House coronavirus spokesperson, moved the goalpost and admitted the goal of COVID vaccines is to provide personal protection only, not to prevent death, or person-to-person transmission. Fauci said he and his colleagues would “settle for … the primary endpoint to prevent clinically recognizable disease.”
“Settle for” could be used when someone cannot afford the house they want, or when their favorite pizza topping is not available, so someone settles for cheese pizza instead of pepperoni. It is hard to imagine the words “settle for” would ever be uttered in reference to a vaccine, let alone by the guy leading the COVID vaccine program for the U.S.
Settling for a vaccine that does not meet the initially lofty promises will not make more people voluntarily line up to get it.
Flawed trial design
As conceded by Fauci, there are indeed some concerning issues with the trial design, spelled out nicely by Dr. Peter Doshi in the British Medical Journal. Doshi focuses on the two biggest issues. First, none of the leading vaccine candidate trials is designed to test if the vaccine can reduce severe COVID-19 symptoms, defined as: hospital admissions, ICU or death. And, second, the trials are not designed to test if the vaccine can interrupt transmission.
If neither of these conditions is met, the vaccine in essence performs like a therapeutic drug, except a vaccine would be taken prophylactically, even by the perfectly healthy, and more than likely carries a higher risk of injury than a therapeutic drug. If this were to be true, then therapeutic drugs would be superior to any COVID vaccine.
Preventing severe symptoms?
Regarding the new Pfizer trial results, Paul Offit, director of the Vaccine Education Center at Children’s Hospital of Philadelphia, said: “I want to know the spectrum of disease that the vaccine prevents. You’d like to see at least a handful of cases of severe disease in the placebo group.”
Though Pfizer stated “the study also will evaluate the potential for the vaccine candidate to … [prevent] against severe COVID-19 disease,” Pfizer’s press release did not indicate if the cases described in the company’s Phase III early release results were mild or severe. “In all the ongoing phase III trials for which details have been released, laboratory confirmed infections even with only mild symptoms qualify as meeting the primary endpoint definition,” wrote Doshi.
Phase III trials include a challenge test, where those who are vaccinated and those in the placebo group are followed to see if they end up testing positive for COVID-19, referred to as events or cases. “Final efficacy analyses are planned after just 150 to 160 ‘events,’” stated Doshi, “regardless of severity of the illness.” He went on to say that “hospital admissions and deaths from COVID-19 are simply too uncommon in the population being studied for an effective vaccine to demonstrate statistically significant differences in a trial of 30,000 people.”
The entire point of clinical trials is to demonstrate statistical significance so the FDA can make an informed decision on whether or not to approve the vaccine. The present clinical trials do not provide reliable data on whether these vaccines prevent hospitalizations and deaths.
No blocking of transmission
Plans to roll out vaccines to the 7.5 billion people in the world based on about 160 clinical trial participants per vaccine candidate not only lacks statistical power; most would consider it reckless.
Regarding the Pfizer results, Mt. Sinai virologist and trial participant Florian Krammer indicated “a transmission-blocking vaccine could accelerate the end of the pandemic. However, it will be difficult to determine if the Pfizer vaccine, or others in late-stage trials, can achieve this.” Moderna Chief Medical Officer Tal Zaks concurs, stating “our trial will not demonstrate prevention of transmission, because … you have to swab people twice a week for very long periods, and that becomes operationally untenable,” citing the need for a five-to-ten times longer trial length and even higher costs.
Since these COVID-19 vaccines will not be approved for full licensure based on their ability to stop the spread of COVID-19 or prevent hospitalization or death, we may face never-ending lockdowns. If the present COVID-19 vaccine clinical trials eventually lead to full licensure, yet do not statistically significantly establish prevention of person-to-person transmission, they should not be used to justify mandated vaccinations in order to board a plane, go to work, attend a concert or eat at a restaurant.
Lack of study power in groups most affected by COVID-19
After Phase I trials, manufacturer studies are recruiting the elderly, minorities and those with underlying health conditions into larger studies with more than 30,000 subjects. However, though Pfizer stated that “approximately 42% of global participants and 30% of U.S. participants have racially and ethnically diverse backgrounds,” there are concerns the trials are not sufficiently powered to evaluate vaccine effectiveness in these groups.
The 160-event challenge tests will not be broken down by sub-population, leaving little actionable data on these vulnerable groups. Additionally, Pfizer has not disclosed how many elderly are enrolled in its trial. “I can’t see how anybody — the DSMB [Data Safety Monitoring Board] or the FDA Vaccine Advisory Committee, or FDA decision-makers — would ever allow a vaccine to be recommended for that group [age 65 and older] without having adequate data,” said Offit.
Adverse events and concerning vaccine ingredients
So exactly what has been revealed thus far by the COVID-19 vaccine trials? Below are the issues with trial design, paused trials, adverse events, and questions about fast-tracking a new mRNA technology, among many other questions and issues.
Here are the five companies hoping the FDA will grant an Emergency Use Authorization: Moderna, Pfizer/BioNTech, Johnson & Johnson, Astra-Zeneca/Oxford, and GlaxoSmithKline. All receive funding from Operation Warp Speed to compress development time into a few months. Normally, vaccines take years to go through proper safety testing.
Significantly, with about two dozen vaccines in active use today, at least 66 formally tested and approved vaccines in the U.S. have been discontinued. Many — like RotaShield (rotavirus), Lymerix, and the DTP shot — were removed due to safety issues. Given the highly compressed development time, the adverse events experienced by trial participants and the possibility of vaccine mandates under full licensure, the public must demand transparency and open access to trial data.
Paused trials
Johnson & Johnson was the latest vaccine maker to pause its COVID-19 vaccine trial due to a severe adverse event in a vaccine recipient. At the Oct. 30 Advisory Committee on Immunization Practices (ACIP) meeting, Johnson & Johnson’s Dr. Jerald Sadoff was pressed by no fewer than six ACIP members to reveal the illness, but refused, citing confidentiality.
Meanwhile, Astra-Zeneca/Oxford also had to pause their trial after trial participants developed neurological conditions like transverse myelitis and multiple sclerosis and due to a death, reportedly in the placebo group, which received meningitis vaccines instead of true saline placebos.
In Moderna’s Phase I trial, at least one participant had to drop out due to urticaria, a common allergic drug reaction that can cause a life-threatening anaphylaxis, but the drugmaker did not pause its trial. Pfizer/BioNTech has stated it will not pause its trial despite “side effects that have emerged.”
On Oct. 23, the FDA cleared both Johnson & Johnson and Astra-Zeneca to resume their trials, stating they could not definitively link the severe adverse events or death to the COVID vaccines. Given the small number of people in the trials, severe adverse events in just a few participants could translate into thousands, if not millions of injuries if the entire world population were vaccinated.
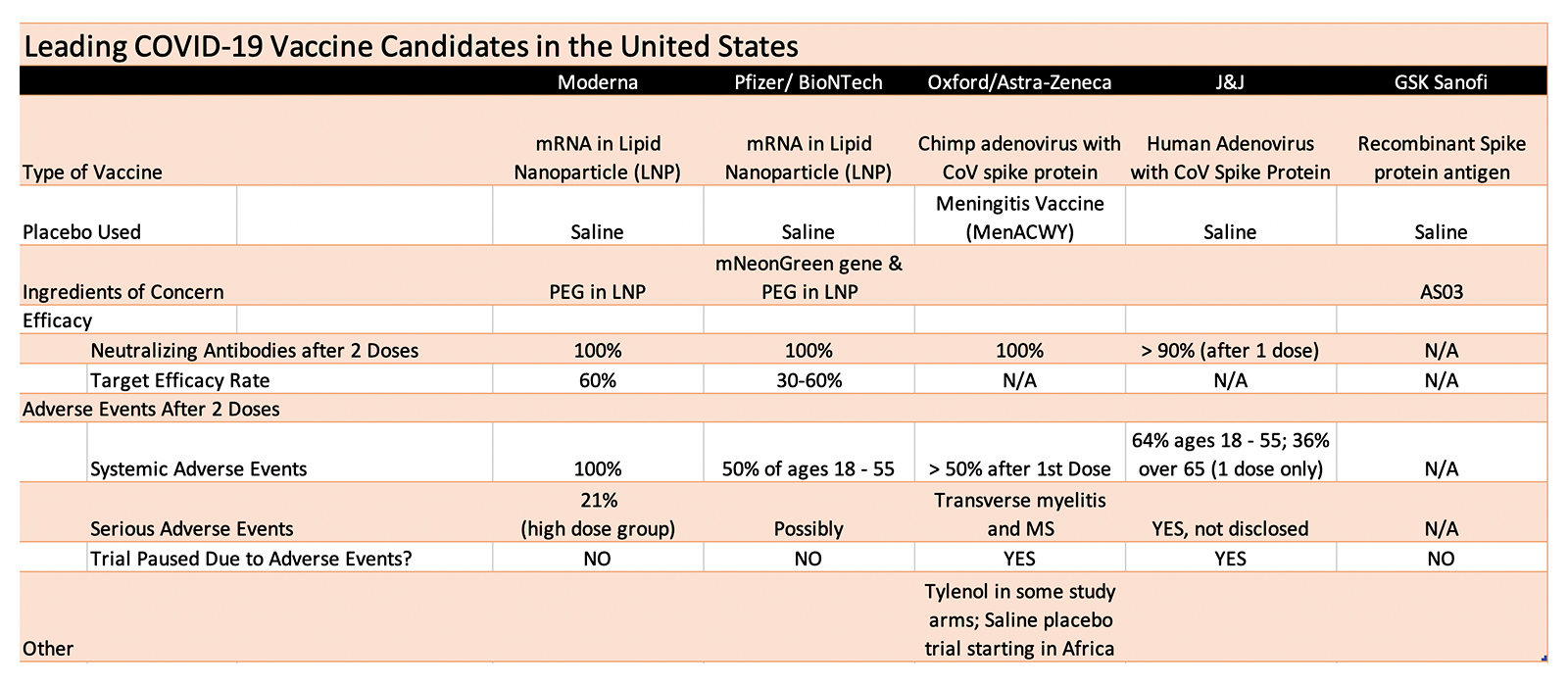
Questionable efficacy
Among the leading COVID vaccine candidates, Moderna, Pfizer/BioNTech, Astra-Zeneca/Oxford and Johnson & Johnson (J&J) have all published data from early human trials. (Links to published studies within the text above, and also cited after the article.)
While all four report 90% to 100% of participants developed antibodies after two doses (single dose for J&J), all four also report a high rate of adverse events. Note that antibodies are merely presumed to be effective, when levels are comparable to antibodies in people who recovered naturally from COVID.
We will not know if the vaccines prevent or reduce symptoms, like the early release results from the Pfizer trial, until complete challenge test results are back. In the challenge test, the vaccinated and those who got a placebo injection remain blinded, that is, they do not know which group they are in. If a participant experiences COVID-like symptoms, he or she will get a “nasal swab” test. Cases will be counted when a participant tests positive for SARS CoV-2, the virus that is thought to cause COVID-19 symptoms, via a polymerase chain reaction test. The vaccinated group will then be compared to the placebo group.
High systemic adverse events
One hundred percent of those injected with two doses of Moderna’s mRNA vaccine (100 mcg) experienced systemic adverse events, while 50% of those aged 18-55 in Pfizer’s trial had systemic adverse events.
In Astra-Zeneca/Oxford’s trial, it took only one dose to cause more than 50% of participants to experience adverse events. In J&J’s trial, a single dose caused almost two-thirds of those under 55 to have systemic adverse events, compared to about a third of those over 65.
Systemic adverse events experienced by participants in all trials include chills, fever, muscle pain and headache, which participants claim last about 24 hours. One man with chills chattered his teeth so badly that he broke a tooth.
mRNA: unproven new vaccine technology
Historically, vaccines are made from an infectious organism — either a virus or a bacterium — that is grown in a cell culture, like egg or aborted fetal cells such as MRC-5.
Vaccine antigens are prepared in four ways: 1) live, but weakened by attenuation, like the measles, mumps, rubella vaccine (MMR); 2) inactivated with a poison such as formaldehyde, like the flu shot; 3) using part of an organism — a subunit, recombinant, polysaccharide or a conjugate vaccine, like Hepatitis B or the shingles vaccine; or 4) using a toxoid (toxin) made by a germ, like a tetanus shot.
In all cases, the resulting vaccine provokes the recipient’s B-cells to make antibodies to that organism. Subunit and toxoid vaccines tend not to work without an adjuvant like aluminum, which causes a more robust immune response.
In a technological departure from the four basic vaccine types, both Moderna and Pfizer/BioNTech are testing mRNA vaccines, a technology that does not appear to rely at all on biological products. mRNA instructs our cells to take action. In the case of the COVID vaccine, the lab works with synthetic mRNA intended to instruct the body’s cellular machinery to make some of the SARS CoV-2 proteins, but not the entire SARS CoV-2 virus. Then the immune system is expected to make antibodies against those parts of the virus. Basically, mRNA vaccines are intended to biohack — through genetic modification — a human being to produce parts of a virus.
Vaccines are classified as biologics, not drugs, because traditional vaccines have always been derived from biological materials. It is questionable that an mRNA vaccine using synthetic RNA — which appears to have nothing biological in it — could still maintain this classification as a biologic. It is really an injected drug, and obviously a huge departure from traditional biologically based vaccine technology. This should give us pause, given the fast-tracked schedule is even more inappropriate for an entirely new vaccine technology.
Autoimmune syndromes caused by vaccination: pathogenic priming and antibody-dependent enhancement
The handful of animal trials performed by the manufacturers — J&J (primate), Moderna (mouse & primate), Pfizer (mouse & primate) and AstraZeneca/Oxford (primate) — focused on overall tolerability of the vaccines, clearance of pathogens from the upper and lower respiratory tract, and probed for which dose level might be immunogenic and safe.
The animal trials are being conducted alongside — not before — human trials, and have yet to release results regarding the possibility of pathogenic priming, which could lead to enhanced COVID-19 disease in individuals vaccinated against the SARS-CoV-2 virus, and potentially cause autoimmunity against many human proteins, including critical proteins in our immune systems.
As far as is known, none of the vaccines has eliminated unsafe epitopes — the part of SARS-CoV-2 proteins that match human proteins. If the immune system produces antibodies to these epitopes, they could attack “self,” the hallmark of autoimmune disease.
We may have to wait for results from Phase III COVID vaccine trials for information on another untoward autoimmune condition, antibody-dependent enhancement (ADE). In ADE, vaccines may cause idiopathic antibodies that act like a Trojan horse for wild viruses, allowing the target virus to enter cells and replicate. The opposite of protecting someone from an infection, ADE causes the vaccinated person to get a worse case of the disease, and possibly suffer organ damage.
A recent review of ADE, also termed immune enhancement, noted current trials are not designed to find ADE, concluding that “rigorous clinical trial design and postlicensure [sic] surveillance should provide a reliable strategy to identify adverse events, including the potential for enhanced severity of COVID-19 disease, after vaccination.”
ADE has been demonstrated in studies on SARS CoV in: humans, ferrets (liver damage) and non-human primates (acute lung damage), among a much larger body of literature.
It is not known what percent of the population may suffer pathogenic priming or antibody-dependent enhancement after vaccination with a COVID vaccine. Estimates of Americans who already have an autoimmune disease range from 14.7 million to 23.5 million. They are likely more susceptible to pathogenic priming and ADE.
Other concerning ingredients
Aside from Moderna and Pfizer/BioNTech, the other leading vaccine manufacturers are using recombinant vaccine technology, producing a genomic chimera with properties intended to both activate the immune system and generate antibodies to the SARS CoV-2 spike protein.
Astra-Zeneca/Oxford is using a chimp virus — an adenovirus — that can be the cause of the common cold, combined with the spike protein from SARS CoV-2. There are long-standing concerns about primate viruses in vaccines ever since the polio vaccine administered from 1955 to 1963 was linked with cancer. The polio vaccine was cultured in primate kidney cells infected with simian virus 40 (SV40).
Johnson & Johnson is using a human adenovirus combined with the spike protein. GlaxoSmithKline/Sanofi is using a recombinant antigen based on their flu vaccine technology.
Veteran vaccine researchers have also raised a warning flag about COVID-19 vaccine candidates that use adenoviruses that could result in an increased susceptibility to HIV infections based on previous findings. In a Lancet report in October, researchers utilizing adenovector COVID vaccine technology acknowledged the “controversial” possibility of their vector increasing the risk of HIV infection, and said they would watch for it in the vaccine candidate trials.
There are some other concerning ingredients to watch closely. In the Moderna and Pfizer mRNA vaccines, polyethylene glycol (PEG) is found in the fatty lipid nanoparticle coating around the mRNA. Seventy percent of people make antibodies to PEG and most do not know it, creating a concerning situation where many could have allergic, potentially deadly, reactions to a PEG-containing vaccine. PEG antibodies may also reduce vaccine effectiveness.
Pfizer is inserting an ingredient derived from a marine invertebrate, mNeonGreen, into its vaccine. The ingredient has bioluminescent qualities, making it attractive for medical imaging purposes, but it is unclear why an injected vaccine would need to have the equivalent of a visual day-glow marker. mNeonGreen has unknown antigenicity.
Finally, the GlaxoSmithKline vaccine will have a well-characterized toxic ingredient, AS03, an adjuvant used in the H1N1 vaccine that was linked with narcolepsy and cataplexy. It contains squalene which is harvested from shark livers, and is linked with Gulf War Syndrome. AS03 also contains polysorbate 80, which disrupts the normally protective blood-brain barrier, and tocopherol, a form of Vitamin E, as an emulsifier.
Meningitis vaccine ‘fauxcebo’
While Pfizer and GSK are using saline placebos in their trials, Astra-Zeneca/Oxford is using a meningitis vaccine as its “placebo,” which some term a “fauxcebo.” The meningitis vaccine causes significant levels of adverse events, and may have even caused the reported death in the Astra-Zeneca/Oxford trial.
Comparing a COVID-19 vaccine to a meningitis vaccine as a placebo may have comparable levels of adverse events, allowing the manufacturers to misleadingly assert their COVID-19 vaccines had no more adverse events than the meningitis placebo. If they actually compared their vaccines to a saline placebo, the COVID-19 vaccine would likely have more adverse events.
In their study protocol, Astra-Zeneca/Oxford stated the “use of saline as a placebo would risk unblinding participants, as those who had notable reactions would know they were in the ChAdOx1 nCoV-19 vaccine group.” Astra-Zeneca/Oxford does have one saline placebo trial planned in South Africa, so there will be safety data compared to a real placebo when that trial is completed in a few years.
Tylenol in some study groups
Finally, another oddity in the Astra-Zeneca/Oxford trial is the use in some study groups of acetaminophen, also known as Tylenol or paracetamol. The vaccine maker explained that it wanted to use the highest vaccine dose possible, so a higher percentage of people would develop immunity after the first dose. Per their Phase I study, “a single higher [vaccine] dose was chosen to provide the highest chance of rapid induction of neutralizing antibody. In the context of a pandemic wave where a single higher, but more reactogenic dose might be more likely to rapidly induce protective immunity, the use of prophylactic paracetamol appears to increase tolerability and would reduce confusion with COVID-19 symptoms that might be caused by short-lived vaccine-related symptoms without compromising immunogenicity.”
Acetaminophen is made from coal tar, and even though it’s been in use since the late 1800s, science is still unsure of its mechanism of action. Side effects are well known, however. It depletes glutathione, the body’s most abundant antioxidant made in the liver. It is a questionable practice to administer this over-the-counter drug with vaccines, as the body needs abundant glutathione to detoxify vaccine ingredients.
BARDA funding and potential approval right around the corner
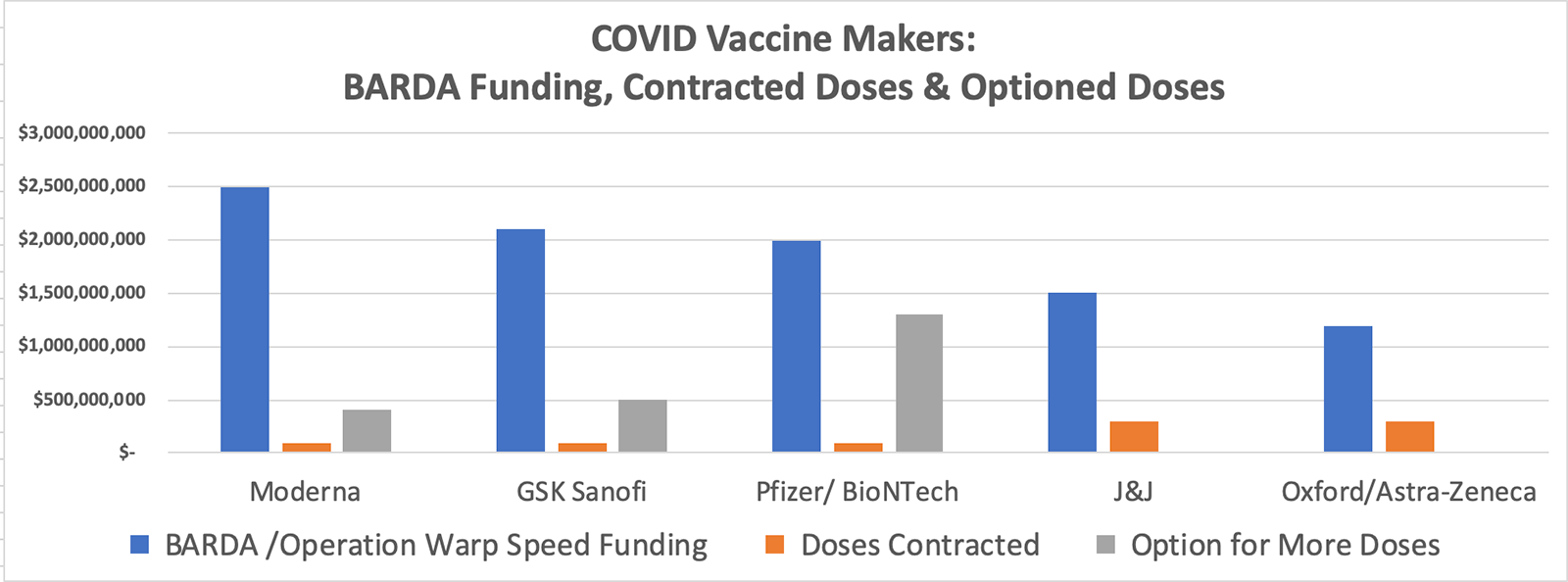
All five of the leading vaccine manufacturers have received money from the Department of Health and Human Services’ Biomedical Advanced Research and Development Authority (BARDA), in amounts ranging from $1.2 to $3 billion to accelerate trials under Operation Warp Speed. Manufacturers are all committed to producing 100 – 300 million initial doses of their vaccines, with contracts to produce millions or billions more doses.
As enthusiasm for a COVID-19 vaccine wanes, it appears the clinical trials will not inspire more confidence. Since everyone eagerly awaits the “new normal” and some think a vaccine is the key to end lockdowns, enthusiasm remains for even a sub-optimal vaccine. If suboptimal means a high rate of serious injury, the vaccine makers still have a long road ahead to prove the vaccines do not cause more death and injury than the symptoms of COVID-19.
Confidence is certainly not boosted when new mRNA vaccine technologies are being tested at Warp Speed led by former GSK executive Moncef Slaoui, who helped conceal Avandia’s severe cardiac adverse events — a clear case of the fox guarding the henhouse.
Trading COVID disabilities and deaths for vaccine injuries and deaths is not an option. Even if manufacturers can show the serious injury rate is less than 1%, if the 7.5 billion people in the world were all vaccinated, millions could be permanently injured or die from the vaccine. Though mandates are prohibited under an Emergency Use Authorization, it will not be too much longer until manufacturers seek full licensure.
However, even under full licensure, if the vaccine doesn’t prevent spread, there is no case to be made for vaccine mandates. Individuals should always have the choice of whether or not to vaccinate themselves or their minor children, after being fully informed of both risks and benefits. As always, Children’s Health Defense awaits a safe and effective vaccine, and opposes all mandated medicines.
Contributors to this article include: James Lyons-Weiler, Ph.D. and Greg Glaser, General Counsel for Physicians for Informed Consent.
Summary of published human trials by manufacturer
Pfizer/BioNTech
Moderna
Astra Zeneca/Oxford
Johnson & Johnson
GSK Sanofi








