Miss a day, miss a lot. Subscribe to The Defender's Top News of the Day. It's free.
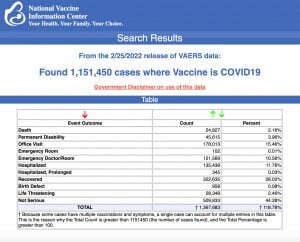
2The Centers for Disease Control and Prevention (CDC) today released new data showing a total of 1,151,450 reports of adverse events following COVID vaccines were submitted between Dec. 14, 2020, and Feb. 25, 2022, to the Vaccine Adverse Event Reporting System (VAERS). VAERS is the primary government-funded system for reporting adverse vaccine reactions in the U.S.
The data included a total of 24,827 reports of deaths — an increase of 425 over the previous week — and 200,331 reports of serious injuries, including deaths, during the same time period — up 4,128 compared with the previous week.
Excluding “foreign reports” to VAERS, 774,373 adverse events, including 11,312 deaths and 74,257 serious injuries, were reported in the U.S. between Dec. 14, 2020, and Feb. 25, 2022.
Foreign reports are reports foreign subsidiaries send to U.S. vaccine manufacturers. Under U.S. Food and Drug Administration (FDA) regulations, if a manufacturer is notified of a foreign case report that describes an event that is both serious and does not appear on the product’s labeling, the manufacturer is required to submit the report to VAERS.
Of the 11,312 U.S. deaths reported as of Feb. 25, 18% occurred within 24 hours of vaccination, 22% occurred within 48 hours of vaccination and 60% occurred in people who experienced an onset of symptoms within 48 hours of being vaccinated.
In the U.S., 552 million COVID vaccine doses had been administered as of Feb. 25, including 325 million doses of Pfizer, 208 million doses of Moderna and 18 million doses of Johnson & Johnson (J&J).
Every Friday, VAERS publishes vaccine injury reports received as of a specified date. Reports submitted to VAERS require further investigation before a causal relationship can be confirmed. Historically, VAERS has been shown to report only 1% of actual vaccine adverse events.
U.S. VAERS data from Dec. 14, 2020, to Feb. 25, 2022, for 5- to 11-year-olds show:
8,817 adverse events, including 200 rated as serious and 4 reported deaths.
The most recent death involves an 8-year-old boy (VAERS I.D. 2109625) from Mississippi who died 7 days after his second dose of Pfizer’s COVID vaccine when he was found blue and lifeless at home.
He was taken to the hospital with a full code in process. A pulse was detected several times, but the boy ultimately died in the ICU. It was reported to the doctor who filed the report that the boy died from multisystem inflammatory syndrome. He did not have COVID.
- 17 reports of myocarditis and pericarditis (heart inflammation).
- 32 reports of blood clotting disorders.
U.S. VAERS data from Dec. 14, 2020, to Feb. 25, 2022, for 12- to 17-year-olds show:
- 29,797 adverse events, including 1,714 rated as serious and 39 reported deaths.
The most recent death involves a 13-year-old girl (VAERS I.D. 2115839) from Wisconsin who was severely compromised and received two doses of Pfizer’s COVID vaccine. Although the cause of death wasn’t clear, she appeared to have significant health issues, respiratory distress and heart problems.
- 69 reports of anaphylaxis among 12- to 17-year-olds where the reaction was life-threatening, required treatment or resulted in death — with 96% of cases
attributed to Pfizer’s vaccine. - 648 reports of myocarditis and pericarditis with 631 cases attributed to Pfizer’s vaccine.
- 159 reports of blood clotting disorders, with all cases attributed to Pfizer.
U.S. VAERS data from Dec. 14, 2020, to Feb. 25, 2022, for all age groups combined, show:
- 19% of deaths were related to cardiac disorders.
- 54% of those who died were male, 41% were female and the remaining death reports did not include the gender of the deceased.
- The average age of death was 72.6.
- As of Feb. 25, 5,176 pregnant women reported adverse events related to COVID vaccines, including 1,653 reports of miscarriage or premature birth.
- Of the 3,587 cases of Bell’s Palsy reported, 51% were attributed to Pfizer vaccinations, 40% to Moderna and 8% to J&J.
- 853 reports of Guillain-Barré syndrome, with 41% of cases attributed to Pfizer, 30% to Moderna and 28% to J&J.
- 2,339 reports of anaphylaxis where the reaction was life-threatening, required treatment or resulted in death.
- 1,621 reports of myocardial infarction.
- 13,320 reports of blood clotting disorders in the U.S. Of those, 5,946 reports were attributed to Pfizer, 4,744 reports to Moderna and 2,568 reports to J&J.
- 4,060 cases of myocarditis and pericarditis with 2,505 cases attributed to Pfizer, 1,373 cases to Moderna and 172 cases to J&J’s COVID vaccine.
Pfizer vaccine only 12% effective in kids 5 to 11, study says
A study released Monday showed the effectiveness of Pfizer’s COVID vaccine in 5- to 11-year-olds was only 12% after a seven-week period of observation. Yet, the authors of the study still recommended the vaccine for that age group, stating it was protective against severe disease.
A study released Monday showed the effectiveness of Pfizer’s COVID-19 vaccine in 5- to 11-year-olds was only 12% after a 7-week period of observation. According to New York Times, federal health officials knew about the findings since early February.https://t.co/yW33Z7JePh
— Robert F. Kennedy Jr (@RobertKennedyJr) March 2, 2022
They also proposed the recommended dose for 5- to 11-year-olds was too small, suggesting a higher dose might solve the problem.
Some scientists at the CDC and FDA pushed for the data to be made public before an FDA meeting, scheduled for Feb. 15, to review Pfizer’s application for Emergency Use Authorization of a three-dose regimen of its vaccine for infants and children 6 months to 5 years old — but the study’s findings were only made public this week.
The FDA on Feb. 11 abruptly postponed the meeting to review Pfizer’s application for infants and children under 5, stating Pfizer didn’t have enough data on the efficiency of a third dose for that age group.
In an article published March 1, Dr. Madhava Setty, senior science editor at The Defender, examined the data in the study and outlined two flaws in the authors’ conclusions: 1) that the vaccines prevent severe disease in this age group, and 2) that increasing the dose might be appropriate.
Judge clears way for Pfizer whistleblower lawsuit
A whistleblower lawsuit alleging fraud during Pfizer’s COVID vaccine trials is moving forward, after a district court judge unsealed the complaint, including 400 pages of exhibits.
A whistleblower lawsuit alleging fraud during Pfizer’s COVID vaccine trials is moving forward, after a district court judge unsealed the complaint, including 400 pages of exhibits.https://t.co/qgNImhN5Ov
— Robert F. Kennedy Jr (@RobertKennedyJr) March 2, 2022
Brook Jackson in January 2021 sued Pfizer and two contractors that worked on its COVID vaccine clinical trials: Ventavia Research Group and ICON PLC.
Jackson worked for Ventavia for a brief period in 2020, before being fired after she filed a complaint with the FDA over alleged indiscretions she observed during the vaccine trials.
She also gave The BMJ a cache of internal company documents, photos and recordings highlighting alleged wrongdoing by Ventavia.
Jackson filed the complaint in the U.S. District Court, Eastern District of Texas, Beaumont Division, under the False Claims Act.
The lawsuit includes several charges of fraud and retaliation on the part of both Ventavia and Pfizer. The complaint remained under seal until Feb. 10, when U.S. District Court Judge Michael Truncale ordered it unsealed.
Husband of woman who died from J&J shot speaks out
The husband of an Oregon woman who died last year from a blood-clotting disorder — two weeks after receiving J&J’s COVID vaccine — spoke out publicly this week about his wife’s death.
U.S. health officials continue to say blood-clotting disorders like one that killed 52-year-old Monica Melkonianare two weeks after the J&J vaccine are rare — despite thousands of vaccine-induced blood-clotting events reported to CDC.https://t.co/QqgqgJY6tE
— Robert F. Kennedy Jr (@RobertKennedyJr) March 2, 2022
Stan Thomas told NBC News he’s fighting to ensure his wife’s sacrifice is not forgotten.
Monica Melkonian, 52, received her J&J shot at a vaccination clinic on April 7, 2021 — the same day the CDC and FDA temporarily paused the vaccine to investigate reports of a rare blood-clotting disorder called vaccine-induced thrombotic thrombocytopenia.
Melkonian’s most notable symptoms included a persistent headache and pain behind her left eye before she experienced a seizure, stroke and ultimately died from the condition.
To date, the U.S. has confirmed only nine deaths due to VITT caused by the J&J shot and 54 cases of blood clotting conditions among J&J recipients, despite thousands of cases reported to VAERS.
Booster shots causing more injuries than expected in Israel
More Israelis are experiencing injuries and reactions following COVID booster shots than the country’s passive reporting system shows, according to a survey conducted by the Israeli Ministry of Health (MOH).
The MOH conducted an active survey of booster shot recipients to collect data on adverse events associated with booster doses, then compared the survey data to the data available from the country’s passive reporting system.
Out of 4,000 people who participated in the survey, results showed six of 2,049 respondents were admitted to the hospital following the booster shot, and a significant number of people reported worsening of their underlying health conditions.
Extrapolated to the millions of booster doses that have been administered, that’s 270,000 hospitalizations per 92 million booster doses administered in the U.S. and 13,000 hospitalizations per 4.5 million booster shots administered in Israel.
The MOH survey also found reports of allergic reactions, menstrual irregularities, neurological injuries, injection-site reactions and general adverse events.
Children’s Health Defense asks anyone who has experienced an adverse reaction, to any vaccine, to file a report following these three steps.