Miss a day, miss a lot. Subscribe to The Defender's Top News of the Day. It's free.
A new study suggests recipients of Moderna’s COVID-19 vaccine may be more likely to suffer repeated infections, perhaps indefinitely.
The study, still in preprint, found participants in Moderna’s adult trial who received the vaccine, and later were exposed to the virus, did not generate antibodies to a key component of the virus as often as did those in the placebo group.
The authors’ findings, which are corroborated by U.K. data that demonstrate the rates of infection are significantly higher in the vaccinated, suggest Moderna knew of this safety signal in 2020 when the vaccine maker was conducting its trials.
The authors of “Anti-nucleocapsid antibodies following SARS-CoV-2 infection in the blinded phase of the mRNA-1273 Covid-19 vaccine efficacy clinical trial“ wrote:
“Among participants with PCR-confirmed Covid-19 illness, seroconversion to anti-N Abs at a median follow up of 53 days post diagnosis occurred in 21/52 (40%) of the mRNA-1273 vaccine recipients vs. 605/648 (93%) of the placebo recipients (p < 0.001).”
Vaccinated participants in the trial who developed breakthrough COVID-19 — meaning they received a positive PCR test — mounted an antibody response to the nucleocapsid portion of the SARS-CoV-2 virus less often than did placebo recipients who tested positive for the virus.
The difference was statistically significant, leading the authors to conclude:
“Vaccination status should be considered when interpreting seroprevalence and seropositivity data based solely on anti-N Ab testing.
“As a marker of recent infection, anti-N Abs may have lower sensitivity in mRNA-1273-vaccinated persons who become infected.”
In other words, the authors found that using the presence of anti-nucleocapsid (anti-N) antibodies to determine whether a person was exposed to SARS-CoV-2 will miss some infections. Thus, the sensitivity of this kind of test, when applied to vaccinated individuals, is not ideal.
However, there are more important implications of these findings, as Igor Chudov and others were quick to recognize.
Specifically, the study implies that the reduced ability of a vaccinated individual to produce antibodies to other portions of the virus may lead to a greater risk of future infections in the vaccinated compared to the unvaccinated.
It is important to note that this is not just another argument for the superiority of natural immunity.
Rather, this is evidence suggesting that even after a vaccinated person has a breakthrough infection, that individual still does not acquire the same level of protection against subsequent exposures that an unvaccinated person acquires.
This is a troubling finding, and something investigators conducting the Moderna vaccine trial likely knew in 2020.
Let’s establish some background facts:
- Anti-nucleocapsid antibodies are antibodies specific to the nucleocapsid portion of the SARS-CoV-2 virus, the virus responsible for COVID-19.
- One method of determining whether an individual has been exposed to the virus (recently or in the past) is by detecting antibodies specific to the virus in the person’s serum.
- Because those who have been vaccinated will, ostensibly, have anti-spike-protein antibodies, using anti-spike levels to diagnose prior or recent infection is impossible in those people.
However the presence of anti-nucleocapsid antibodies after exposure should, in theory, be unaffected by vaccination status. - Though the presence of an anti-N antibody is not necessarily indicative of immunity, having a broad set of antibodies to different components of the virus offers more robust protection than having antibodies to a single component, e.g. the spike protein.
This is one reason natural immunity is superior to mRNA vaccine-mediated immunity. - The paper does not offer any new findings. Instead it presents an analysis of what should have already been known from Moderna’s vaccine trial.
Breakthrough infections result in N-antibody production less often than in primary infection
Moderna enrolled more than 30,000 subjects in its trial, randomizing them equally to the vaccine or placebo.
At the end of the initial observation period, 13 times more placebo recipients contracted COVID-19, allowing the investigators to boast a Vaccine Efficacy (VE) of 93.2%.
The U.S. Food and Drug Administration (FDA) subsequently authorized the vaccine for emergency use. The rest is history.
This recent study examined the presence or absence of anti-nucleocapsid antibodies in all trial participants who contracted COVID-19, based on a positive PCR test, during the trial period.
As stated earlier, the placebo recipients produced anti-N antibodies more than twice as often as their counterparts who were vaccinated. (Chudov offers a good explanation of the significance of anti-N antibodies here.)
This is a puzzling finding. Why would vaccination suppress a person’s ability to produce antibodies to different parts of the virus when exposed to the entire virus?
One possibility is that if the vaccine is protective, viral loads will be lower, leading to fewer seroconversions.
However, this study also found that at any given viral load, the unvaccinated produced a larger anti-N response than the vaccinated:
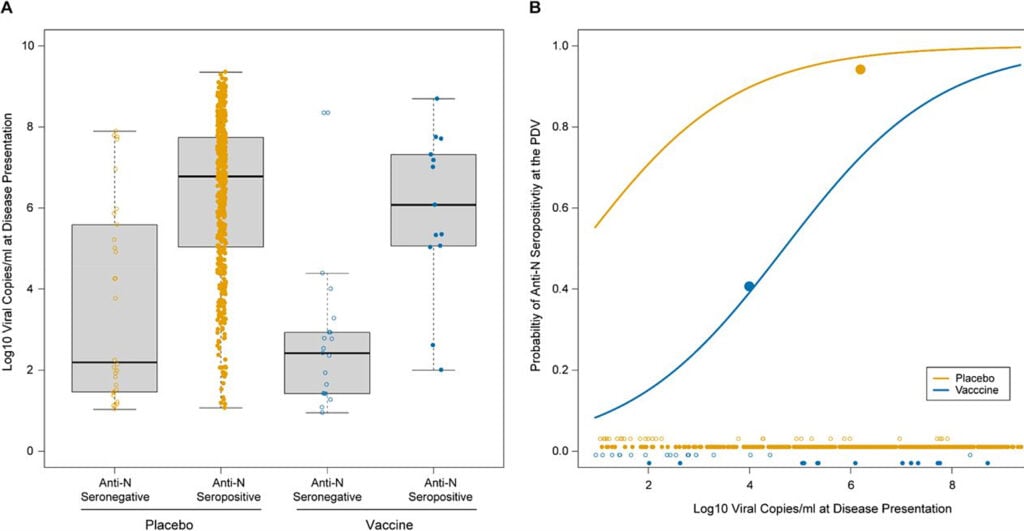
The plot on the left (A) shows the number of placebo and vaccine recipients who seroconverted after exposure to SARS-Co-V2 at varying levels of viral copies. We can see that regardless of vaccination status, lower viral copy numbers result in fewer seroconversions than at higher viral copy numbers.
But there is a significant difference between the vaccinated and placebo recipients.
This is demonstrated in Plot B, which shows that for any given viral copy number, the placebo recipients were significantly more likely to seroconvert. The difference is most prominent at lower viral copy numbers.
This is not a spurious finding. The UK Health Security Agency publishes weekly surveillance data and monitors the levels of anti-N antibodies in their blood donors.
They noted the lower levels of these antibodies in vaccinated individuals who had breakthrough infections. However, they attributed this lower level to the protective benefit of the vaccine.
According to a March 22 report by the U.K. agency:
“These lower anti N responses in individuals with breakthrough infections (post-vaccination) compared to primary infections likely reflect the shorter and milder infections in these patients.”
Though vaccination may for some time decrease severity of the disease, “shorter and milder” (less severe) infections are associated with lower viral load.
As the data above demonstrate, the vaccinated have a lower anti-N antibody conversion rate than the unvaccinated at all levels of viral load.
This is one of the most significant findings of the study because it overturns the heretofore unchallenged idea that decreased seroconversion in the vaccinated is due to less severe infection in this population — which is a benefit provided by the vaccine.
However, this new study shows that even at low viral loads, the unvaccinated are more likely to seroconvert than those who are vaccinated.
In fact, the difference in seroconversion rates is the greatest at lowest viral loads.
The decrease in conversion rates is not a result of a benefit from the vaccine. It is a consequence of it.
Once vaccinated, there may be no going back
The authors also uncovered another important finding: Participants in the Moderna trial who were PCR-positive and seronegative at baseline, prior to inoculation with vaccine or placebo, eventually seroconverted equally — independent of whether they received the vaccine or placebo.
This was also true in participants who received only one dose.
This means that vaccination status at the time of exposure is predictive of seroconversion rate in the future, and the effect is dose-dependent.
Whether or not seroconversion results in more robust immunity is yet to be determined. However, data from the U.K. and elsewhere suggest lower conversion rates may explain why infection rates are higher in their vaccinated population. This data is presented below.
Will repeated bouts of COVID-19 eventually lead to seroconversion in the vaccinated? The study could not answer this question.
More importantly, how will regular boosting affect seroconversion rates as time goes on? This is another important question yet to be answered.
There is no question that vaccine-mediated immunity against COVID-19 wanes and is waning faster as time goes on. The Centers for Disease Control and Prevention (CDC) recommended a first booster after five months and a second after only four.
Israeli data surrounding the effectiveness of a second booster demonstrated that effectiveness in preventing “severe disease” (not hospitalization) fell to just above 50% within seven weeks.
If a third booster is offered, it will probably be recommended sooner than four months after the second, based on this disappointing data.
There is more disappointing data coming from the U.K. In their last weekly surveillance report, which includes case rates between the unvaccinated and boosted citizens, the Health Security Agency offered this disquieting table:
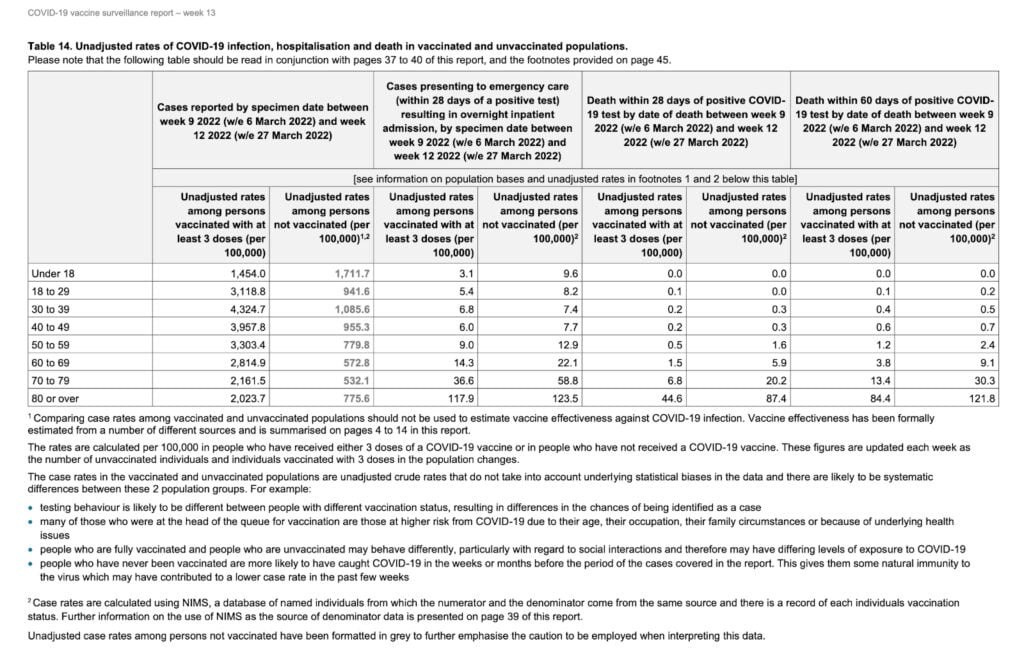
The first two columns demonstrate that the COVID-19 case rate in the boosted was three to four times higher than in the unvaccinated in all ages except for under 18.
The agency warns us to interpret these numbers with caution. Vaccinated people may demonstrate less cautious behavior than the unvaccinated. And there is also no risk stratification based on comorbidities.
However, the chart does account for the biggest factor — age.
So what could explain such a large increase in infection rates among the boosted?
Interestingly, the authors further warn that the unvaccinated may have contracted COVID-19 prior to the observation period — in other words, they may have acquired natural immunity previously, giving them added protection.
But shouldn’t this apply to the boosted as well? It should, but the authors don’t mention that.
Because it is not mentioned, we can safely say the authors assume the unvaccinated are more likely than the boosted to have succumbed to a previous infection. Thus, there will be more people in the unvaccinated pool enjoying the advantage of natural immunity.
But their own data tells the opposite story. The boosted are more likely to contract the disease — by a factor of 3 to 4. How do we know whether the larger infection rates in the boosted are due to more robust immunity in the unvaccinated because of prior infection or due to an immune deficiency in the boosted?
The question can be definitively answered by examining the trend of infection rates. Here is the equivalent table from two months earlier:
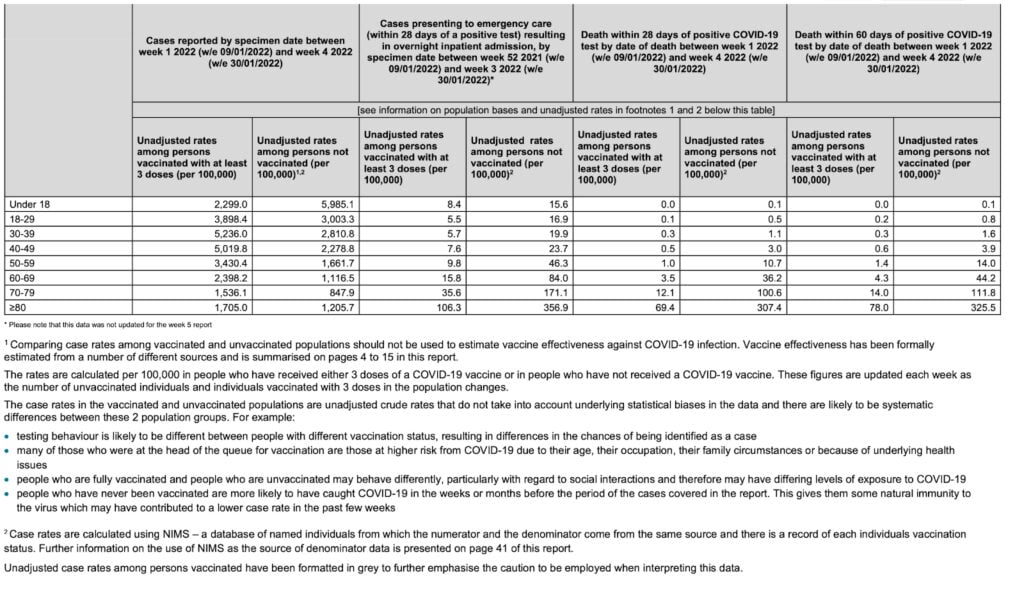
There is still a greater infection rate among the boosted, but it is only two to three times higher. If the authors’ hypothesis was correct, the more recent data should have shown less of a difference, not more.
The growing spread between the vaccinated and unvaccinated is a trend that has been in effect for months. Curious readers can examine the data for themselves here.
If anything, their data support the finding that the decreased seroconversion rates in the vaccinated may be causing a greater risk of repeated infections.
However, without knowing how many of the infections in the boosted are second or third bouts of COVID-19, we can only speculate.
The U.K. data are not outliers. Latest data from Walgreens further demonstrate the boosted are getting infected at nearly twice the rate of the unvaccinated.
Unfortunately, the Walgreens data also do not specify how many of these infections are reinfections.
How many people have already been exposed or infected?
The CDC on April 29 released its latest Morbidity and Mortality Weekly Report (MMWR) titled, “Seroprevalence of Infection-Induced SARS-CoV-2 Antibodies — United States, September 2021–February 2022.”
In its report, the CDC estimates the percentage of the population that has been previously infected with SARS-CoV-2. The agency determined this through a seroprevalence study looking for anti-N antibodies in serum collected from more than 45,000 people from 52 different jurisdictions.
Based on the percentage of anti-N antibodies in serum samples, estimates are that 57.7% of the U.S. population has been exposed to SARS-CoV-2 as of February.
However, this estimate does not consider the fact that the vaccinated do not seroconvert at the same rate as those who are unvaccinated.
This percentage is significantly higher than COVID-19 cases that have been reported to date. As of April 24, there were approximately 70.5 million cases, which amounts to only 21.4% of the population (estimated at 330 million).
The reason for this difference is that seroprevalence studies will pick up all cases and exposures, including in the asymptomatic. In that sense, seroprevalence studies will offer a more accurate estimate of the portion of the population that has been exposed.
Notably, seroprevalence varied significantly between age groups: 75.2% of sera from children ages 0-11 were positive for anti-N antibodies, compared to 33.2% in people 65 and older.
The CDC did not report on the seroprevalence by vaccination status. Instead the agency acknowledged its findings “might underestimate the cumulative number of SARS-CoV-2 infections because infections after vaccination might result in lower anti-N titers.”
Interestingly, the CDC authors cited the very same preprint study discussed above to explain this possibility.
What does this mean for children?
Though the CDC inconveniently chose not to report seroconversion rates in the vaccinated versus the unvaccinated, we can still make some reasonable and important estimations from this weekly report based on our recent understanding of seroconversion rates in the vaccinated.
As stated above, the CDC reported that serum samples drawn from children ages 11 and under tested positive for anti-N antibodies 75.2% of the time.
A central question is, what portion of the seropositive samples were found in the unvaccinated?
Though the FDA does not consider seropositivity a surrogate for immunity, knowing whether or not a child has been exposed to SARS-CoV-2 will be useful when weighing the risk versus benefit of vaccination.
Vaccine uptake is only 28.6% in ages 5 to 11. Children under 5 are ineligible for vaccination. If we assume that few, if any, children under 5 received the COVID-19 vaccine, only 16.4% of children under 12 have been vaccinated based on population data in that age group.
If 16.4% of children under 12 have been vaccinated, the unvaccinated outnumber the vaccinated by approximately 5 to 1. If the vaccinated seroconvert as frequently as the unvaccinated we would expect that one-sixth of the seropositives came from the vaccinated and five-sixths came from the unvaccinated.
However, data from the Moderna trial demonstrate that the vaccinated convert only 40% of the time compared to 93% in the unvaccinated. The vaccinated seroconvert (40/93) = 0.43 times as often as the unvaccinated.
We can then say that the ratio of vaccinated seropositives to unvaccinated seropositives is 0.43 to 5, or 1 to 11.63. This means that 1/12.63, or 7.9% of the seropositives are from vaccinated children.
The remaining 92.1% (11.63/12.63) are from unvaccinated children.
Thus if 75.2% of sera from children are testing positive for anti-N antibodies, 68.4% (75.2% x 0.921) of unvaccinated children have already seroconverted.
There are potential errors in this calculation:
- The samples used in the survey may not be representative of the population with regard to vaccination status.
- Seroconversion rates among the vaccinated may be higher than what was found in the moderna trial.
- Seroconversion rates may be different in people vaccinated with a different vaccine than Moderna (Pfizer, in this case).
- Seroconversion rates may be different in children.
Nevertheless, we can say with reasonable confidence that a substantial proportion of unvaccinated children have already been exposed to SARS-CoV-2.
With known risks of adverse events that now include a reduced ability to generate anti-N antibodies after exposure, there is no justification for requiring or even recommending the vaccine to children without assessing their antibody levels first.
Though the FDA continues to maintain that antibody levels are not necessarily indicative of immunity, it nonetheless granted Emergency Use Authorization (EUA) for Pfizer’s product in children ages 5 to 11 based on antibody responses after vaccination.
Summary
Sixteen months have passed since the FDA granted Moderna EUA for its vaccine. More than 200 million doses of this product have been given to people in the U.S. alone.
Did the vaccine manufacturer know in 2020 that its product could potentially impede recipients from mounting broad antibody responses after exposure to SARS-CoV-2?
Does this potential adverse effect occur in other mRNA COVID-19 “vaccines”?
Vaccinated individuals are at increased risk for contracting COVID-19 compared to the unvaccinated.
But what happens after a breakthrough infection? Have these products permanently mitigated our ability to fight off future SARS-CoV-2 infections?
With the majority of children having been already exposed, is there any reason to inoculate even one healthy child before these questions are answered definitively?
Yet the vaccine manufacturer is pushing the FDA to authorize its product for children under age 6.
If these inoculations prevent people from acquiring proper immunity after exposure, they are, in fact, creating a pandemic of the vaccinated.
Less than 10 months ago, CDC Director Dr. Rochelle Walensky warned, “There is a clear message that is coming through. This is becoming a pandemic of the unvaccinated.”
Which is more ironic? Her statement? Or that the statement is coming from an organization called the Centers for Disease Control and Prevention?







