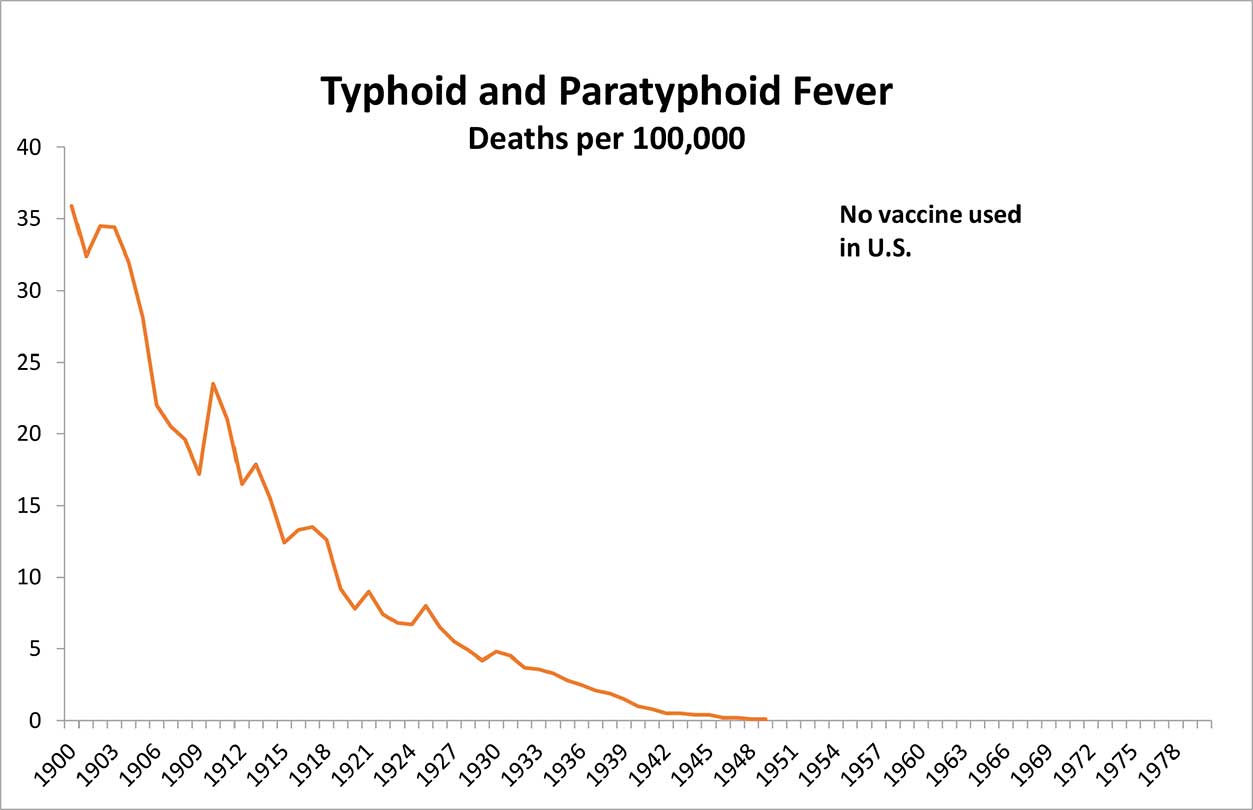
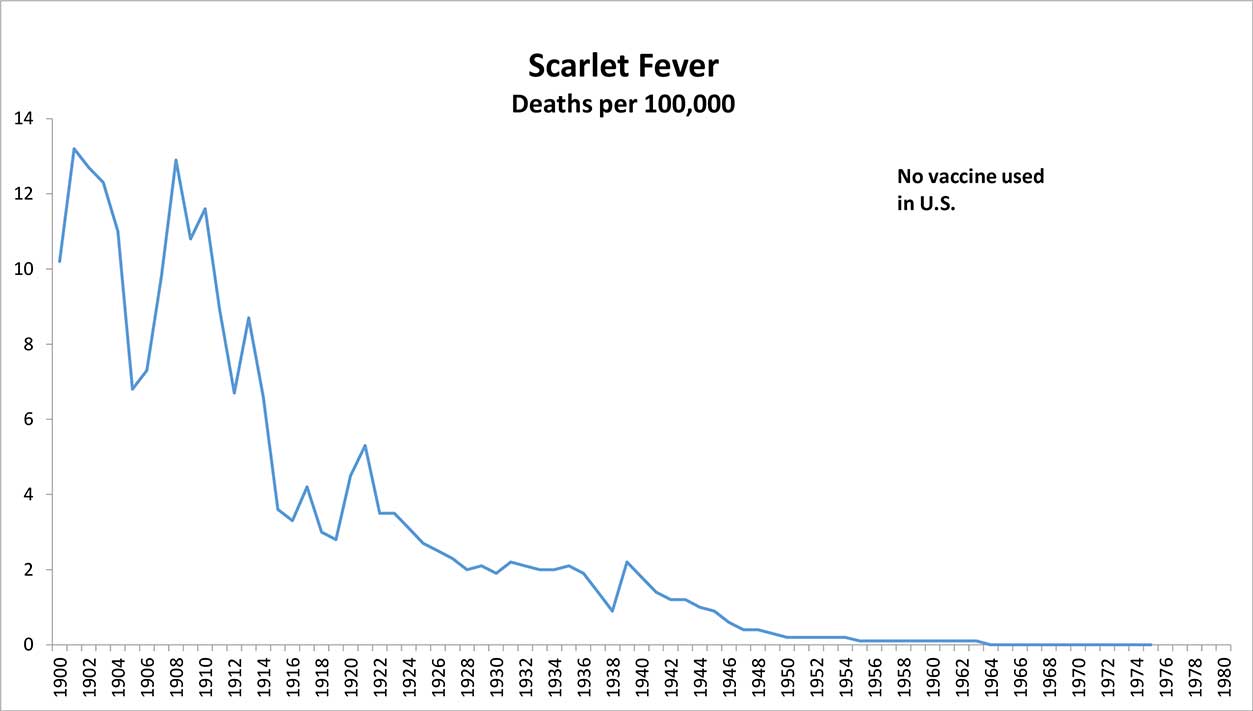
No Legal Requirement to Improve Vaccines
Vaccine manufacturers are under no legal requirement and have no financial incentive to improve their vaccines. This is primarily a legal decision by the government. Here’s some of the history:
In 1986, Congress enacted the National Childhood Vaccine Injury Act (NCVIA) against the backdrop of a wave of tort litigation that threatened to drive manufacturers out of the business of providing vaccines. Children were getting severely injured from vaccines. Vaccine makers were getting sued. The vaccine makers went to Congress and basically said if you don’t protect us from this liability, we will stop making vaccines.
Vaccine Makers Protected From Liability
The NCVIA was passed and contained a number of provisions. It created the Vaccine Injury Compensation Program (VICP) and liability protection for vaccine makers so they could not be sued for certain actions.
The question before the U.S. Supreme Court in the case of Bruesewitz v. Wyeth LLC, 562 U.S. 223 (2011) was whether the NCVIA stops vaccine makers from being sued when their vaccine injures or kills someone.
For you legal eagles, the question before the Supreme Court was whether the NCVIA preempts all design-defect claims against vaccine manufacturers brought by plaintiffs seeking compensation for injury or death caused by a vaccine’s side effects.
A design defect occurs when there is an inherent flaw in a product’s design that renders it unreasonably dangerous.
With drugs and almost all other products, it is possible to sue for “design defect.” This basically means that you can sue if the manufacturer sells you a product that is not safely designed. Whether this was no longer possible was being decided in this case in respect to vaccines.
Bruesewitz Decision
In this case, Hannah Bruesewitz got the DTP vaccine when she was 6 months old (April 1992). Within 24 hours, she started to experience seizures. Hannah suffered over 100 seizures during the next month, and her doctors eventually diagnosed her with “residual seizure disorder” and “developmental delay.” Hannah, a teenager at the time the case was brought forward, was still diagnosed with both conditions.
The court’s opinion was written by Justice Scalia. In it he said a number of things including that a vaccine’s design “is not open to question” and that “the safest design is not always the best one.”
Part of the analysis rested on the question of what was “unavoidable?” The statute said that a manufacturer is not liable for hurting a child if the side effect could not be avoided (was “unavoidable”).
42 USC 300aa-22.
The statute states:
“(b) Unavoidable Adverse Side Effects; Warnings.—
“(1) No vaccine manufacturer shall be liable in a civil action for damages arising from a vaccine-related injury or death associated with the administration of a vaccine after the effective date of this subtitle if the injury or death resulted from side effects that were unavoidable even though the vaccine was properly prepared and was accompanied by proper directions and warnings.
DTP Vaccine Approved in 1948
The vaccine at issue was a DTP vaccine manufactured by Lederle Laboratories. It first received federal approval in 1948 and received supplemental approvals in 1953 and 1970. Remember, Hannah was vaccinated in April, 1992 so this vaccine had remained virtually unchanged for 44 years.
During that 44 year span, newer and arguably safer DTP vaccines had or could have been invented.
In front of the U.S. Supreme Court, Hannah’s lawyers argued that when there is a safer vaccine available, a manufacturer must act reasonably in pursuing that safer design. This was an important point because there was no law that required a vaccine maker to make a vaccine in the most efficacious way with the least serious harm to children. This was not a legal requirement.
In addition, neither the CDC nor the FDA were (or are) legally required to do a comparative analysis for safety that is provided under State law design defect claims. The defendant (the drug maker with support from the U.S. Government, Solicitor General, Department of Justice, for the United States) disagreed.
Supreme Court Decision
The opinion written by Justice Scalia also stated:
“A side effect of a vaccine could always have been avoidable by use of a differently designed vaccine not containing the harmful element…We have no idea how much more expensive an alternative design can be before it “compromis[es]” a vaccine’s cost or how much efficacy an alternative design can sacrifice to improve safety.”
Supreme Court Dissent
The dissent was written by Justices Sotomayor and Ginsburg and stated in part:
“[T]he majority’s decision leaves a regulatory vacuum in which no one—neither the FDA nor any other federal agency, nor state and federal juries—ensures that vaccine manufacturers adequately take account of scientific and technological advancements. This concern is especially acute with respect to vaccines that have already been released and marketed to the public. Manufacturers, given the lack of robust competition in the vaccine market, will often have little or no incentive to improve the designs of vaccines that are already generating significant profit margins. Nothing in the text, structure, or legislative history remotely suggests that Congress intended that result. (emphasis added)
What Does All This Mean?
What all this means is that vaccine manufacturers are under no legal requirement and have no financial incentive to improve their vaccines.
It’s “unavoidable” that their products will have side effects and if the vaccine was made any safer it may not work as well. Plus they don’t have to pay if their vaccine injures a child. This is the only group of products in the U.S. that get this special treatment.
The court decided that people may not sue a vaccine maker for failing to update the vaccine with a newer, safer version. By protecting vaccine makers in this way, the government has taken away a vaccine maker’s incentive to develop improved designs. It’s an immense giveaway to the vaccine industry and it means that older vaccines will probably never be changed or significantly improved despite all the new advancements and technology that exists today that didn’t when the vaccine was made decades ago.