Miss a day, miss a lot. Subscribe to The Defender's Top News of the Day. It's free.
The Centers for Disease Control and Prevention (CDC) today released new data showing a total of 1,237,647 reports of adverse events following COVID vaccines were submitted between Dec. 14, 2020, and April 15, 2022, to the Vaccine Adverse Event Reporting System (VAERS). VAERS is the primary government-funded system for reporting adverse vaccine reactions in the U.S.
The data included a total of 27,349 reports of deaths — an increase of 373 over the previous week — and 222,836 serious injuries, including deaths, during the same time period — up 2,971 compared with the previous week.
Excluding “foreign reports” to VAERS, 807,242 adverse events, including 12,566 deaths and 80,170 serious injuries, were reported in the U.S. between Dec. 14, 2020, and April 15, 2022.
Foreign reports are reports foreign subsidiaries send to U.S. vaccine manufacturers. Under U.S. Food and Drug Administration (FDA) regulations, if a manufacturer is notified of a foreign case report that describes an event that is both serious and does not appear on the product’s labeling, the manufacturer is required to submit the report to VAERS.
Of the 12,566 U.S. deaths reported as of April 15, 16% occurred within 24 hours of vaccination, 21% occurred within 48 hours of vaccination and 59% occurred in people who experienced an onset of symptoms within 48 hours of being vaccinated.
In the U.S., 568 million COVID vaccine doses had been administered as of April 15, including 335 million doses of Pfizer, 214 million doses of Moderna and 19 million doses of Johnson & Johnson (J&J).
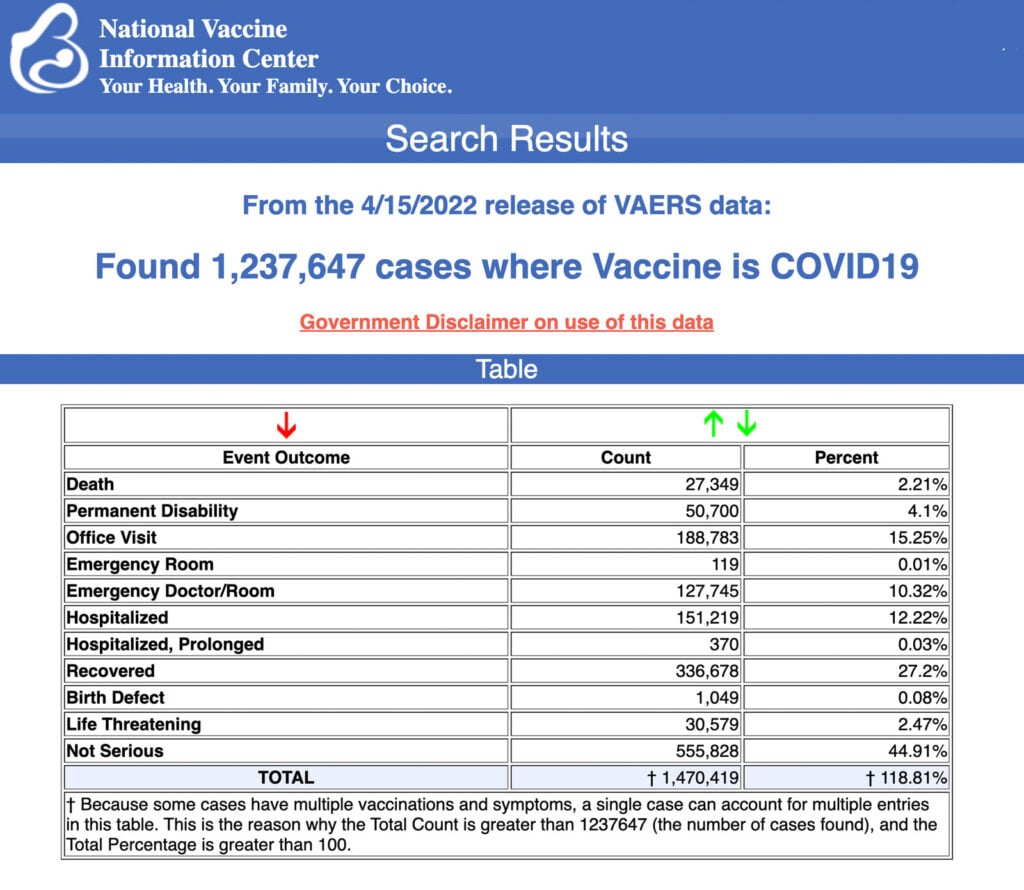 Every Friday, VAERS publishes vaccine injury reports received as of a specified date. Reports submitted to VAERS require further investigation before a causal relationship can be confirmed.
Every Friday, VAERS publishes vaccine injury reports received as of a specified date. Reports submitted to VAERS require further investigation before a causal relationship can be confirmed.
Historically, VAERS has been shown to report only 1% of actual vaccine adverse events.
U.S. VAERS data from Dec. 14, 2020, to April 15, 2022, for 5- to 11-year-olds show:
- 10,290 adverse events, including 248 rated as serious and 5 reported deaths.
- 19 reports of myocarditis and pericarditis (heart inflammation).
The CDC uses a narrowed case definition of “myocarditis,” which excludes cases of cardiac arrest, ischemic strokes and deaths due to heart problems that occur before one has the chance to go to the emergency department. - 40 reports of blood clotting disorders.
U.S. VAERS data from Dec. 14, 2020, to April 15, 2022, for 12- to 17-year-olds show:
- 31,113 adverse events, including 1,796 rated as serious and 43 reported deaths.
- 65 reports of anaphylaxis among 12- to 17-year-olds where the reaction was life-threatening, required treatment or resulted in death — with 96% of cases attributed to Pfizer’s vaccine.
- 651 reports of myocarditis and pericarditis, with 639 cases attributed to Pfizer’s vaccine.
- 166 reports of blood clotting disorders, with all cases attributed to Pfizer.
U.S. VAERS data from Dec. 14, 2020, to April 15, 2022, for all age groups combined, show:
- 20% of deaths were related to cardiac disorders.
- 54% of those who died were male, 41% were female and the remaining death reports did not include the gender of the deceased.
- The average age of death was 73.
- As of April 15, 5,429 pregnant women reported adverse events related to COVID vaccines, including 1,701 reports of miscarriage or premature birth.
- Of the 3,633 cases of Bell’s Palsy reported, 52% were attributed to Pfizer vaccinations, 40% to Moderna and 8% to J&J.
- 861 reports of Guillain-Barré syndrome, with 42% of cases attributed to Pfizer, 30% to Moderna and 28% to J&J.
- 2,355 reports of anaphylaxis where the reaction was life-threatening, required treatment or resulted in death.
- 1,672 reports of myocardial infarction.
- 13,733 reports of blood-clotting disorders in the U.S. Of those, 6,166 reports were attributed to Pfizer, 4,914 reports to Moderna and 2,653 reports to J&J.
- 4,131 cases of myocarditis and pericarditis with 2,532 cases attributed to Pfizer, 1,408 cases to Moderna and 181 cases to J&J’s COVID vaccine.
1 in 10 surveyed doctors believe COVID shots aren’t safe
One in 10 primary care physicians don’t agree COVID vaccines are safe, and 9.3% don’t believe the vaccines are effective, according to a survey published in the April issue of Vaccine.
Researchers at the Texas A&M School of Public Health asked 625 primary care physicians whether they strongly agreed, somewhat agreed, neither agreed nor disagreed, somewhat disagreed or strongly disagreed with the following three statements:
- The COVID vaccines are safe.
- The COVID vaccines are effective.
- The COVID vaccines are important.
The results showed that 10.1% did not agree the vaccines were safe, 9.3% did not agree the vaccines were effective and 8.3% did not agree the vaccines were important. Approximately 5.2% of physicians remained unvaccinated at the end of the survey.
The survey was conducted between May 14 and May 25, 2021.
According to Dr. Madhava Setty, given what has transpired over the past 11 months with vaccine injuries, waning effectiveness and the FDA’s efforts to withhold Pfizer data, the number of physicians who believe COVID shots aren’t safe is likely higher.
769 athletes collapsed this year during competition
More than 769 athletes have collapsed on the field during a game from March 2021 to March 2022. The statistic was revealed by One America News Network (OAN), which also found the average age of the athletes who suffered cardiac arrest is just 23 years old.
The unprecedented surge in cardiac arrest and other heart issues among elite athletes coincides with the rollout of COVID vaccines.
Pearson Sharp of OAN said:
“How many 23-year-old athletes were collapsing and suffering heart attacks before this year? Do you know any 23-year-old people who had heart attacks before now? And these are just the ones we know about.
“How many have gone unreported? Nearly 800 athletes — young, fit people in the prime of life — falling down on the field. In fact, 500% more soccer players in the EU are dropping dead from heart attacks than just one year ago.
“Coincidence? When the Pfizer vaccine is known to cause heart inflammation? No. In fact, many doctors treating these players list their injuries and deaths as being directly caused by the vaccine … This is not a coincidence.”
In an updated report by Good Sciencing, a team of investigators, news editors, journalists and “truth seekers” detailed 942 cardiac arrests and other serious issues among athletes, including 620 deaths, following COVID vaccines.
CDC meets to ‘mull over’ next steps for COVID boosters
The CDC’s vaccine advisory committee met Wednesday to “mull over” what’s next for COVID boosters and consider “upgrades” to the vaccines, The Defender reported.
Some members of the Advisory Committee on Immunization Practices suggested “entirely different vaccine formulations could be needed.” Currently, additional booster doses are recommended only for certain people with weakened immune systems and adults 50 and older.
Vaccine effectiveness is unacceptably low, according to James Lyons-Weiler, Ph.D. And given the findings of Jacques Fantini, a biochemist and professor of virology at Aix-Marseille Université in Marseille, France, the vaccines could have negative effectiveness, indicating disease enhancement.
Sara Oliver, one of the CDC’s epidemic intelligence service officers with the Division of Viral Diseases, said the evolution of the virus will be an important consideration for considering “platforms” for future COVID vaccinations.
This is the CDC putting the word out that a second round of vaccine development is expected — and the closest we’ll ever see to the CDC admitting the vaccination program has flopped, Lyons-Weiler wrote.
Novavax announces early data on COVID and influenza combo vaccine
Vaccine maker Novavax on Wednesday announced that early data on its combination vaccine targeting COVID and influenza showed the vaccine produced a strong immune response.
Chief Medical Officer Filip Dubovsky, during a call with reporters, said the Maryland-based company’s early phase clinical trial found that up to 25 micrograms of the COVID formulation combined with up to 35 micrograms of the flu formulation triggered a promising level of protective antibodies in the Novavax COVID-Influenza Combination Vaccine.
Participants in the Phase 1 trial had a median age of 59 and all previously had received COVID vaccines. Novavax plans to move forward with a Phase 2 trial this year to confirm the appropriate dosing levels, and plans to launch a Phase 3 trial on efficacy during the 2023 flu season at the earliest, Dubovsky said.
Novavax’s COVID vaccine does not use mRNA technology but synthesizes the virus spike outside the human body.
The genetic code for the spike is put into a baculovirus that infects insect cells, which then produce copies of the spike that are purified and extracted. The spike copy is then injected into people to induce an immune response against the virus.
The vaccine also uses a novel adjuvant that contains a purified extract from the bark of a tree in South America, to induce a broader immune response.
The World Health Organization in December 2021 approved Novavax’s COVID vaccine (not the new COVID-flu combination vaccine) for use in the European Union, but the FDA has yet to grant Emergency Use Authorization of any Novavax COVID vaccine in the U.S.
Children’s Health Defense asks anyone who has experienced an adverse reaction, to any vaccine, to file a report following these three steps.